



CAD EYE (EW10-EC02)
Endoscopy system
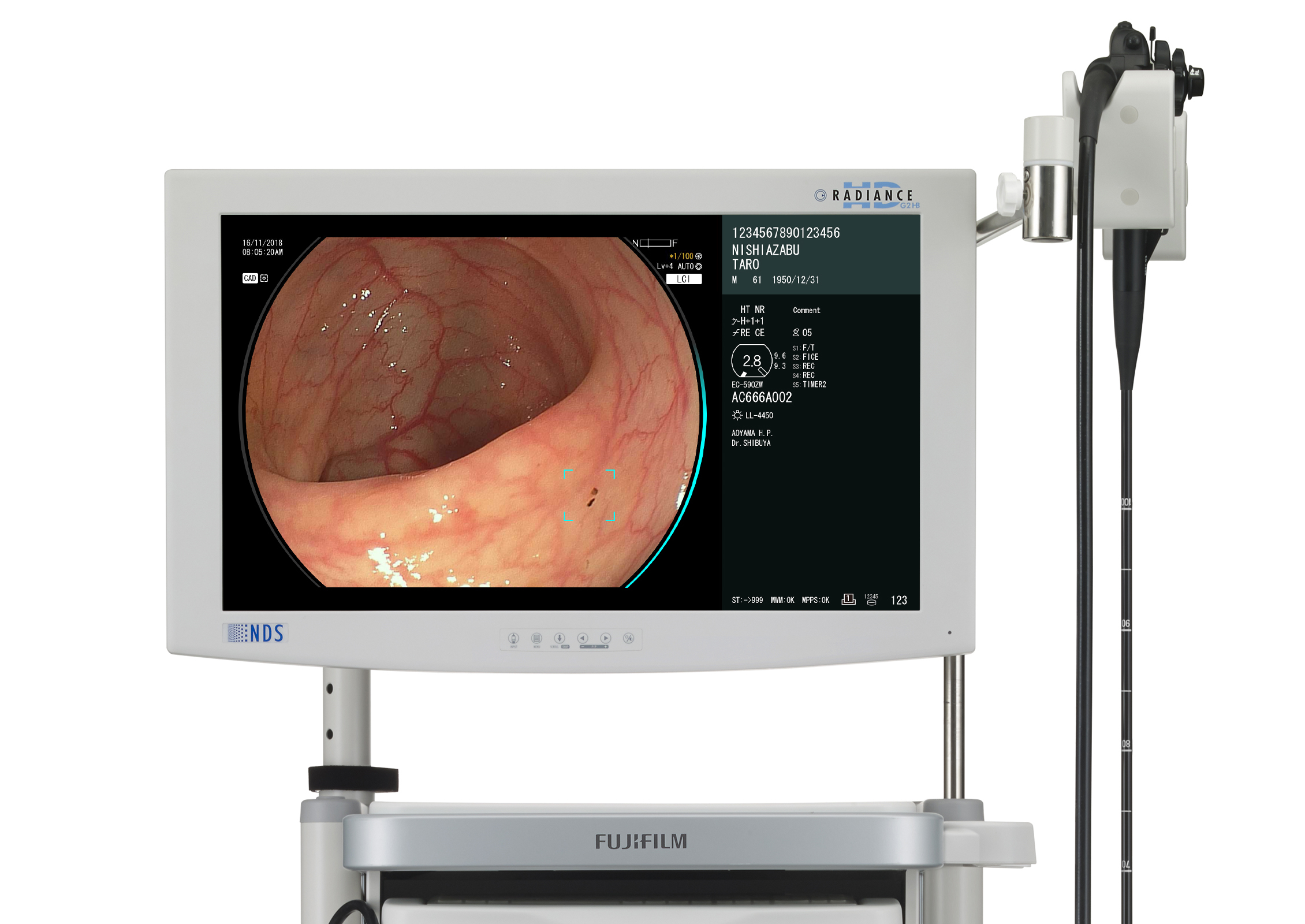
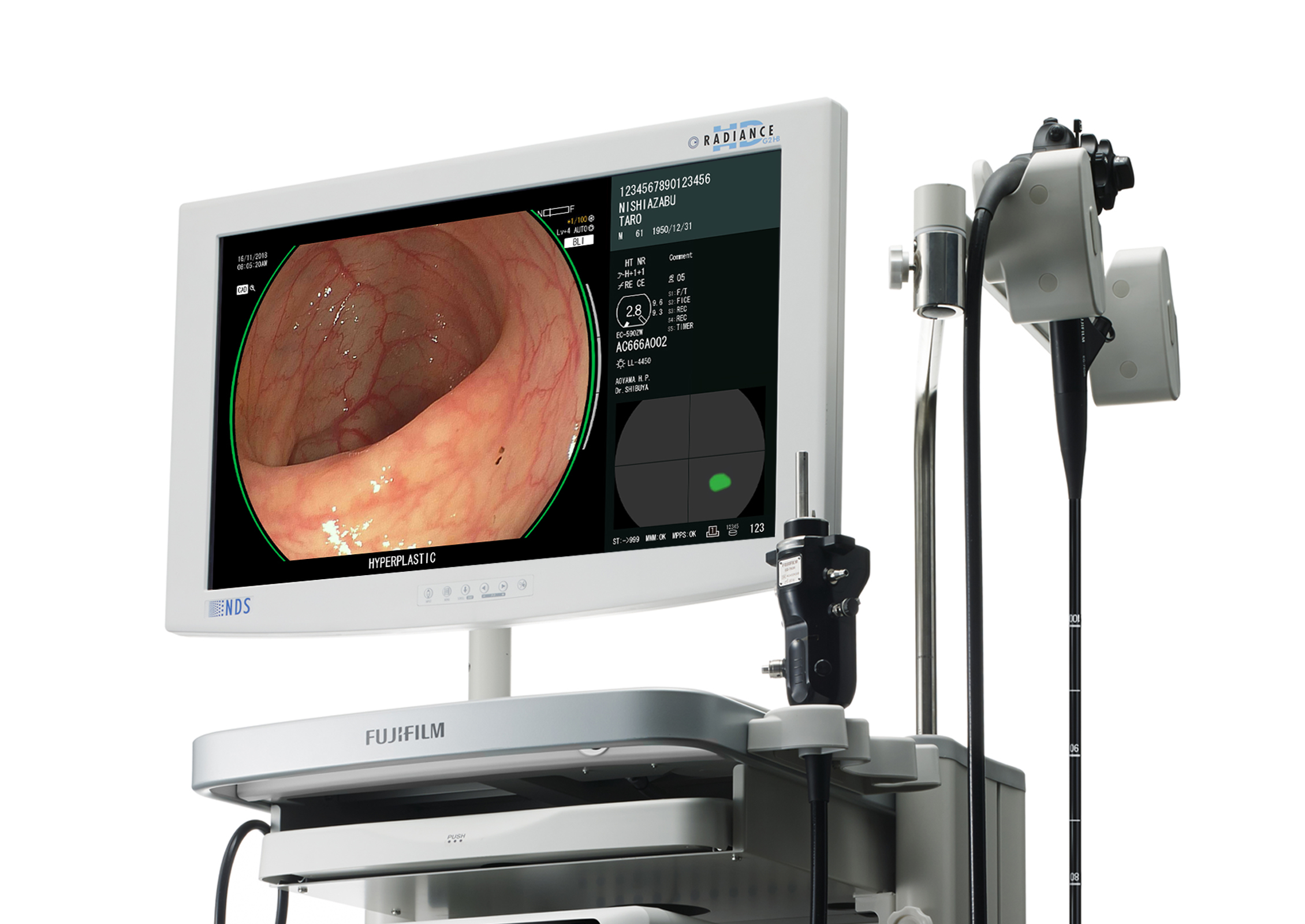
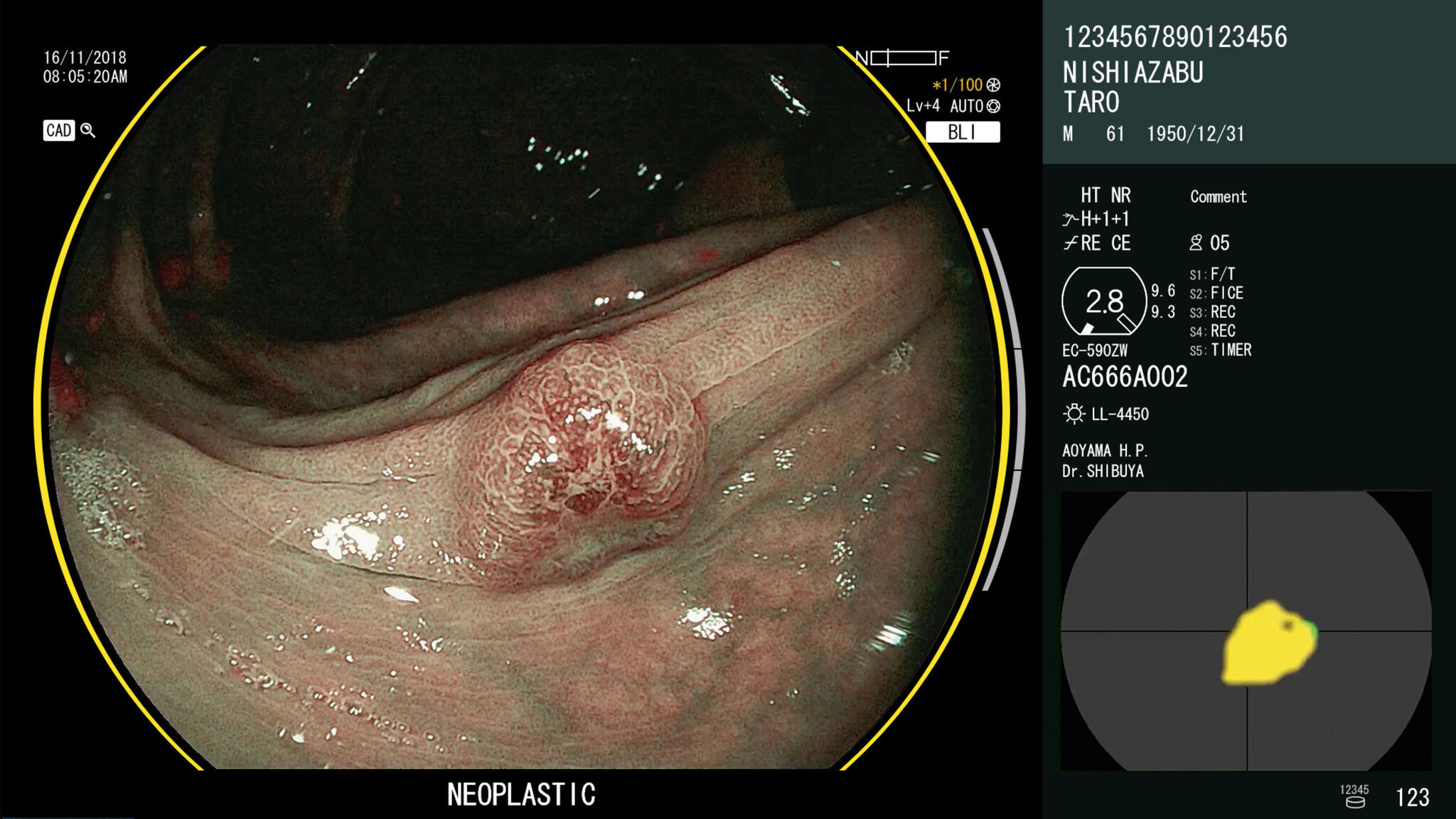
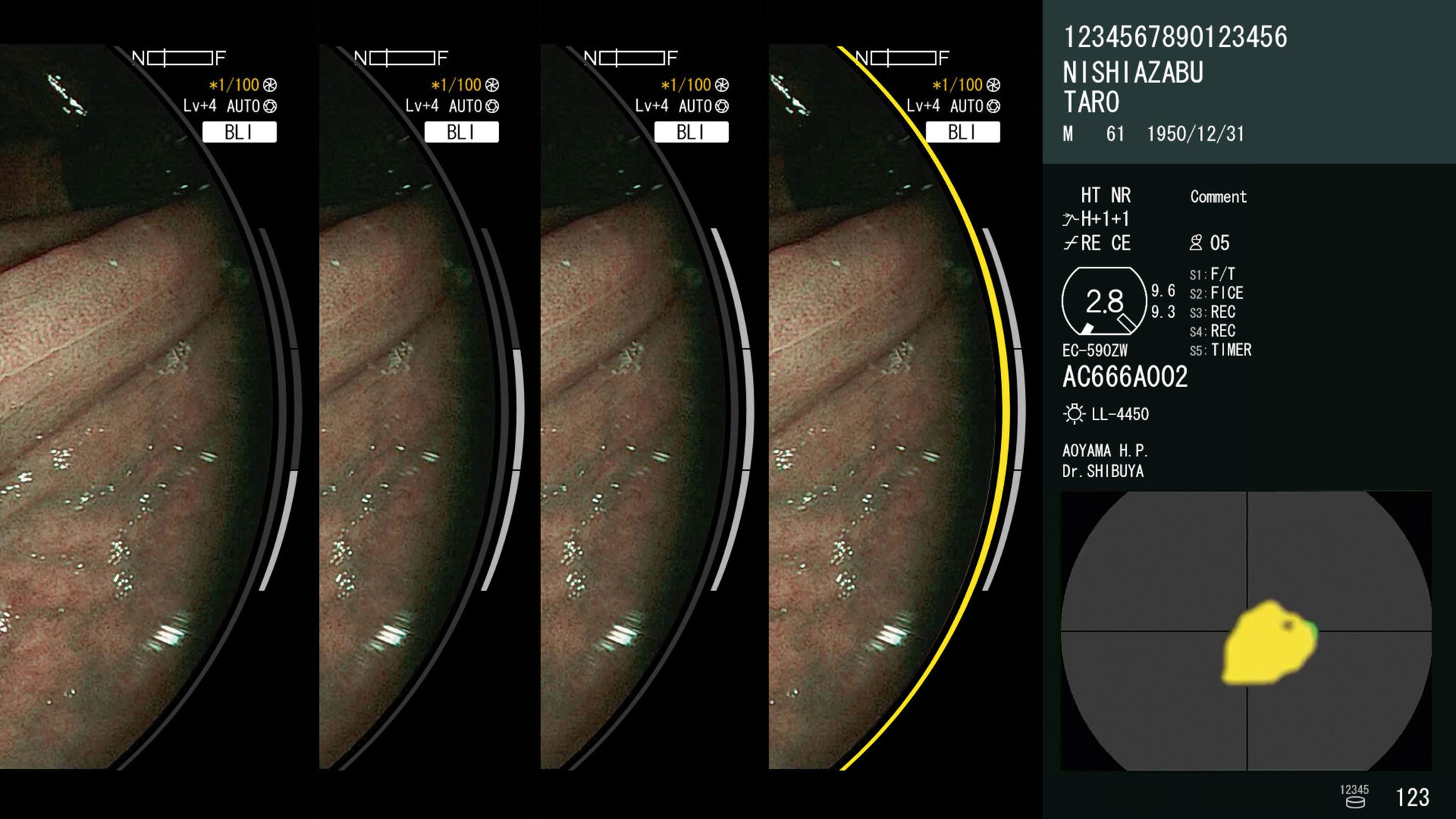
FUJIFILM CorporationCAD EYE is a diagnostic support system that supplements existing endoscopy equipment with AI-powered software. The system analyzes endoscopic images in real-time to detect lesions and can distinguish between hyperplastic and neoplastic polyps. Endoscopists use live images to detect lesions only millimeters in size on the thin, wrinkled inner wall of the large intestine. Such a challenge makes it essential that the imaging technology does not reduce visibility or distract the viewer in any way. The system adds two lines to the available space around the image, which results in an unobtrusive design that better communicates information and supports an accurate diagnosis.
Client / Manufacturer Design
Design

FUJIFILM Corporation
Minato-ku, Tokyo, JP
FUJIFILM Corporation
Minato-ku, Tokyo, JPDate of Launch
2020
Development Time
25 - 36 months
Target Regions
Asia, Europe, North America
Target Groups
Consumer / User, Specific sub-group: medical